To stay up to date on the latest news about Intellia’s technology, pipeline, data releases, and congress presence, sign up to receive email updates from Intellia Medical Affairs.
Want More? Connect With Us!
To stay up to date on the latest news about Intellia’s technology, pipeline, data releases, and congress presence, sign up to receive email updates from Intellia Medical Affairs.
Scientific Exploration in Hereditary Angioedema
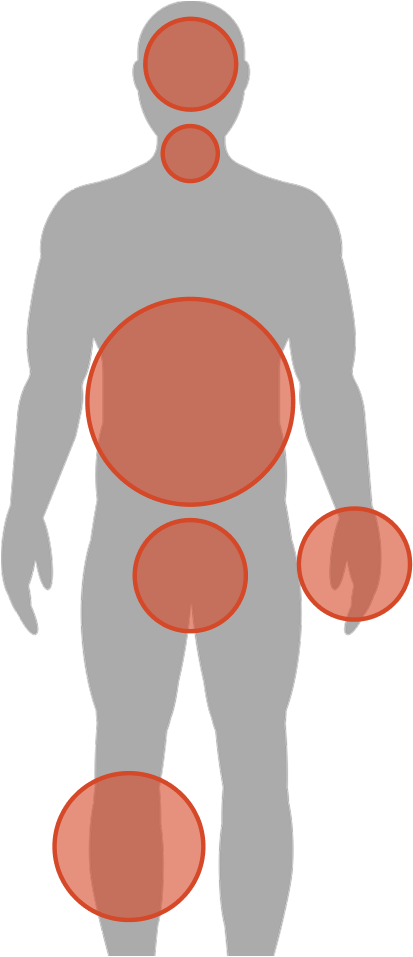
About Hereditary Angioedema
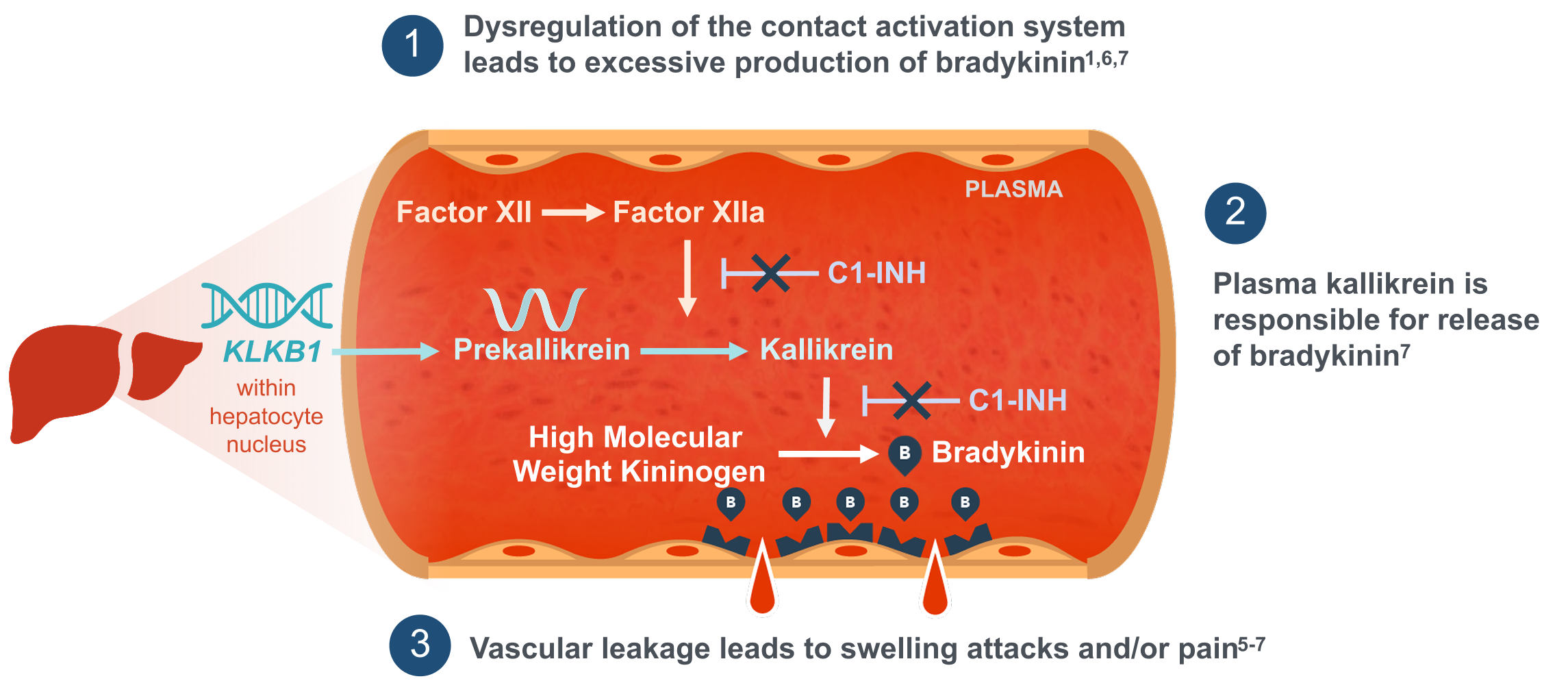
B, bradykinin; C1-INH, C1 esterase inhibitor; KLKB1, kallikrein B1 gene that encodes plasma prekallikrein.
Therapeutic Landscape
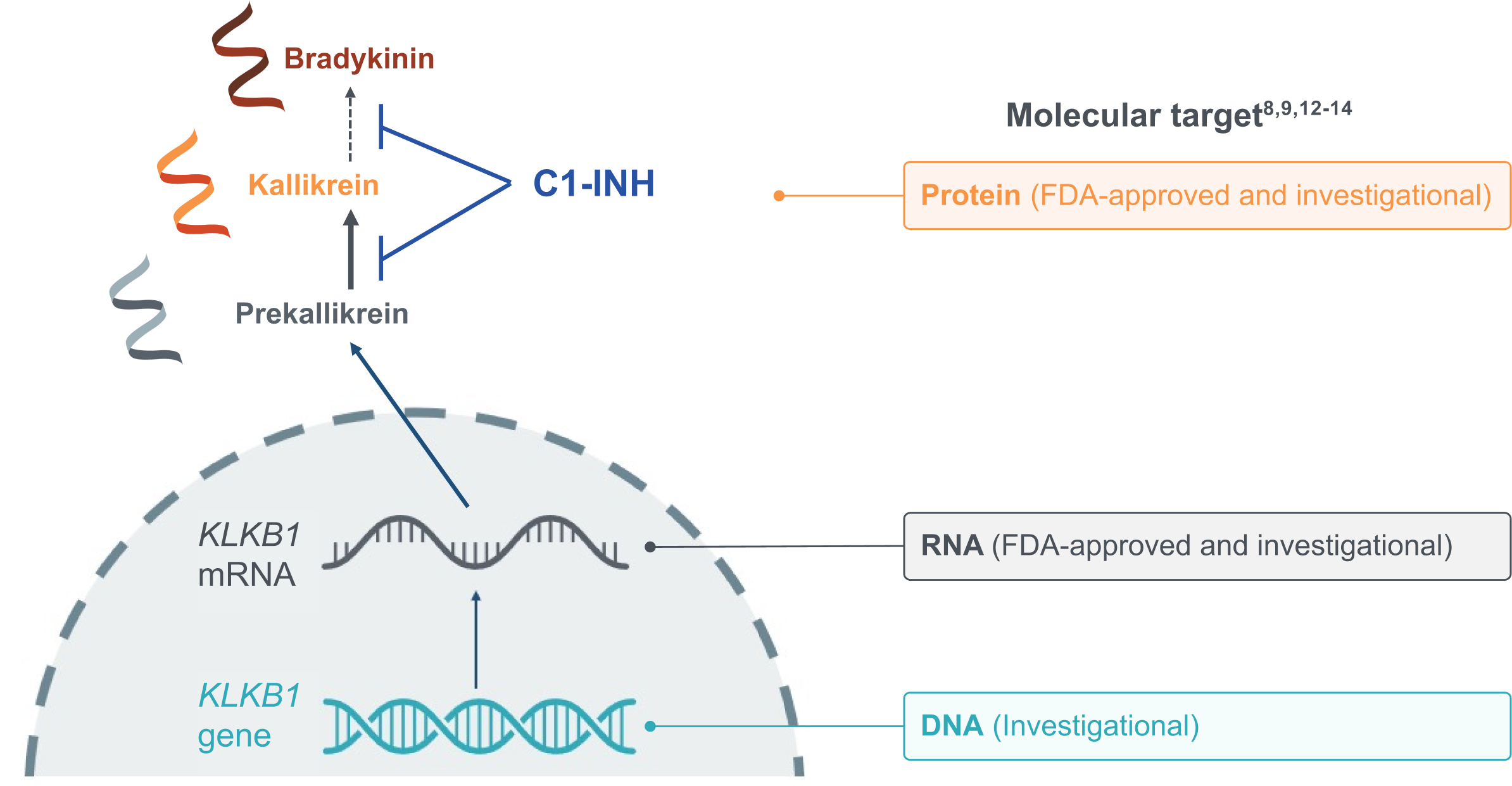
C1-INH, C1 esterase inhibitor; DNA, deoxyribonucleic acid; FDA, US Food and Drug Administration; KLKB1, kallikrein B1 gene that encodes plasma prekallikrein; mRNA, messenger RNA; RNA, ribonucleic acid.
Rationale for CRISPR Research
Connect With Intellia
Our goal is to equip healthcare professionals with the essential knowledge to understand the science of CRISPR and its potential as a therapeutic option to support informed decision-making.
Please submit any specific questions here:
References
- Longhurst HJ, et al. N Engl J Med. 2024;390(5):432-441.
- Bork K, et al. J Allergy Clin Immunol. 2012;130(3):692-697.
- Farkas H. Allergy Asthma Clin Immunol. 2010;6(1):19.
- Mendivil J, et al. Orphanet J Rare Dis. 2021;16(1):94.
- De Maat S, et al. J Thromb Haemost. 2018;16(9):1674-1685.
- Wedner HJ. Allergy Asthma Proc. 2020;41(suppl 1):S14-S17.
- Maurer M, et al. Allergy. 2022;77(7):1961-1990.
- Busse PJ, et al. J Allergy Clin Immunol Pract. 2021;9(1):132-150.e3.
- Donidalorsen. Package insert. Ionis Pharmaceuticals; 2025.
- Press release. KalVista Pharmaceuticals. June 18, 2024. Accessed March 12, 2025. https://ir.kalvista.com/news-releases/news-release-details/kalvista-submits-new-drug-application-fda-sebetralstat-first
- Press release. Pharvaris. December 6, 2023. Accessed March 12, 2025. https://ir.pharvaris.com/news-releases/news-release-details/pharvaris-announces-positive-top-line-phase-2-data-chapter-1
- Smith TD, Riedl MA. Ann Allergy Asthma Immunol. 2024;133(4):380-390.
- Garadacimab-gxii. Package insert. CSL Behring, Inc; 2025.
- Christiansen SC, Zuraw BL. Allergy Asthma Proc. 2020;41(suppl 1):S26-S29.
- Wang D, et al. Nat Rev Drug Discov. 2019;18(5):358-378.
- Drouet C, et al. Front Allergy. 2022;3(835503):1-17.
- Ren Z, et al. J Allergy Clin Immunol. 2023;151(4):1040-1049.e5.
- Longhurst HJ, et al. Supplementary appendix. N Engl J Med. 2024;390(5):432-441.