To stay up to date on the latest news about Intellia’s technology, pipeline, data releases, and congress presence, sign up to receive email updates from Intellia Medical Affairs.
Want More? Connect With Us!
To stay up to date on the latest news about Intellia’s technology, pipeline, data releases, and congress presence, sign up to receive email updates from Intellia Medical Affairs.
Introduction to Genetic Medicine
Therapeutic and investigational strategies to treat life-threatening diseases have advanced over time.¹ Innovation has progressed from small molecule drugs and biologics to RNA- and DNA-targeted therapies.²
Genetic Medicine Delivery Approaches
RNA Therapies

RNA therapies target a specific mRNA, not DNA, to decrease expression of a target protein in a patient.4,5 This approach has demonstrated effectiveness in various diseases, with treatment regimens typically involving dosing intervals ranging from 1 to 6 months.6,7
DNA, deoxyribonucleic acid; mRNA, messenger RNA; RNA, ribonucleic acid.
Gene Therapy
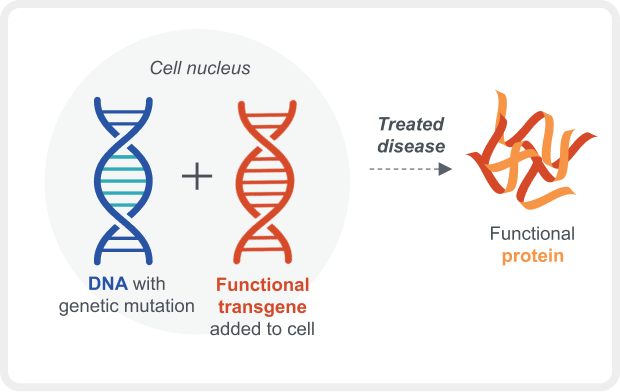
Gene therapy introduces a modified gene into the nucleus of cells with the goal of partially restoring expression of a functional protein.3
With gene transfer, the modified gene is typically delivered via a viral vector, such as adeno-associated virus (AAV), which has been shown to be effective for various diseases.8
DNA, deoxyribonucleic acid.
Gene Editing
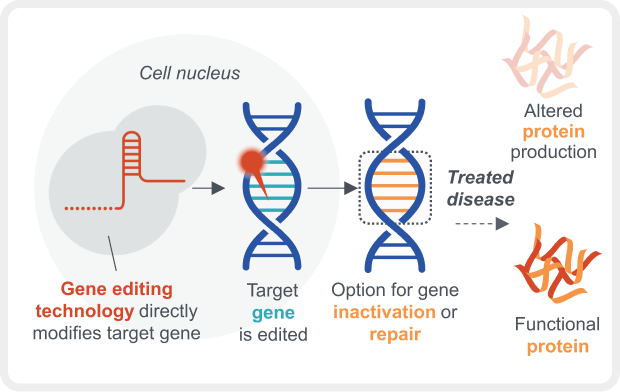
DNA, deoxyribonucleic acid; mRNA, messenger RNA; RNA, ribonucleic acid.
Why CRISPR?
Potential to Revolutionize Treatment of Diseases
Investigational CRISPR-Based Therapies Have the Potential to9,10:

DNA, deoxyribonucleic acid.
Dr. Jim Januzzi, a cardiologist, shares how he communicates the potential outcomes of gene editing with CRISPR to his patients.
Introduction to CRISPR
Editing a Gene Could Alter Production of a Protein Involved in Disease13-17

Since its discovery in 1987, CRISPR has been an established gene editing method studied and used in a variety of applications.20,21
Cas9, CRISPR-associated protein 9; DNA, deoxyribonucleic acid.
Methods of Gene Editing With CRISPR
Inactivate
Insert

Repair
In Vivo
CRISPR can be delivered directly into a patient’s body, also known as in vivo CRISPR.9,10 Investigational in vivo CRISPR therapies are being studied as a one-time intravenous infusion for real-time targeted gene editing.9,10 Depending on the investigational therapy, the in vivo approach may require some premedication, and is intended to be administered in the outpatient setting within a day, with subsequent follow-up and monitoring.23,24
CRISPR, clustered regularly interspaced short palindromic repeats; IV, intravenous.
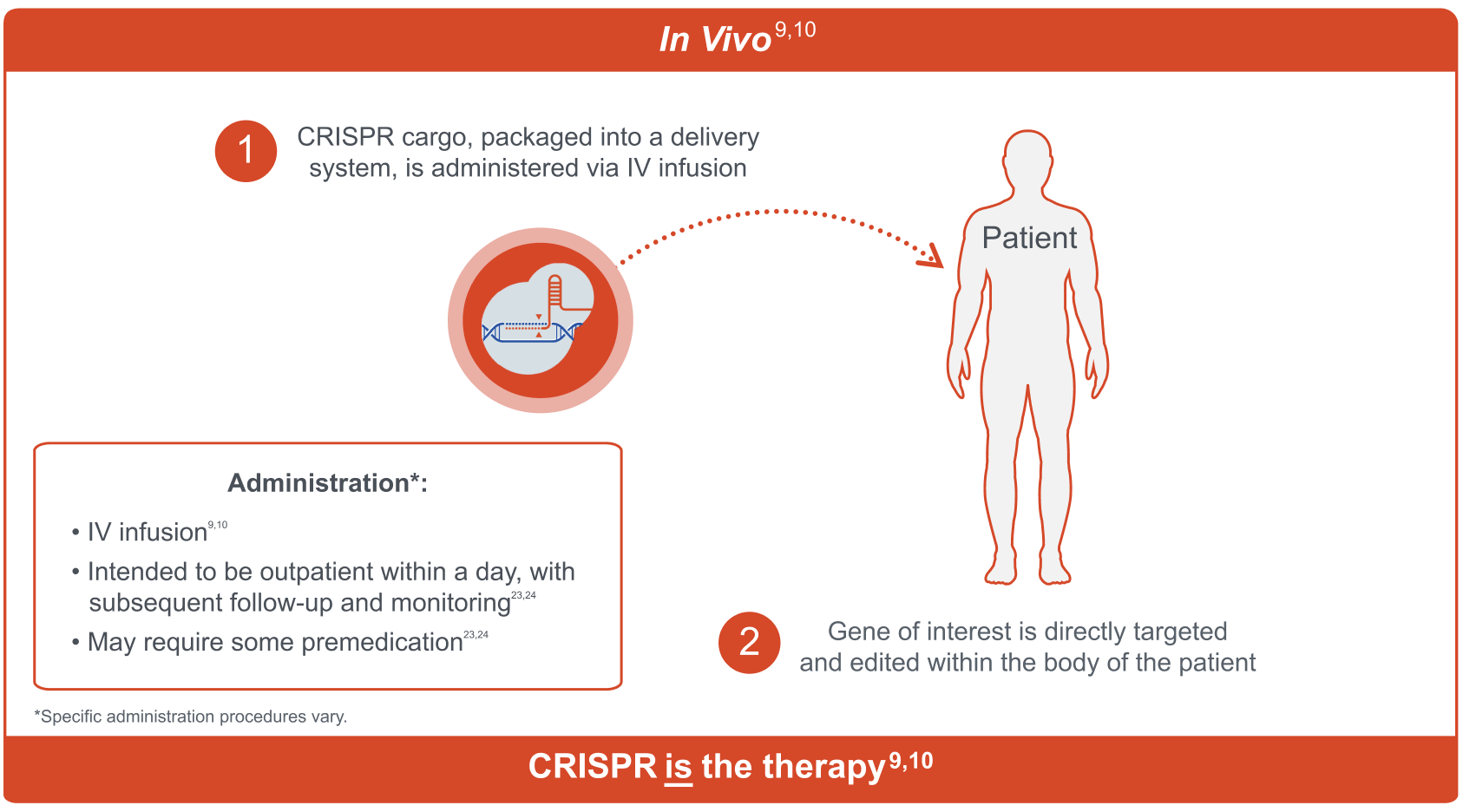
CRISPR, clustered regularly interspaced short palindromic repeats; IV, intravenous.
Ex Vivo
CRISPR can be used to create a therapy outside a patient’s body, also known as ex vivo CRISPR.25 This involves retrieving patient or donor cells, editing them using CRISPR, and then reintroducing them into the patient.25 This method can take months, requires hospitalization, and necessitates immunosuppression prior to therapy.26
CRISPR, clustered regularly interspaced short palindromic repeats; IV, intravenous.
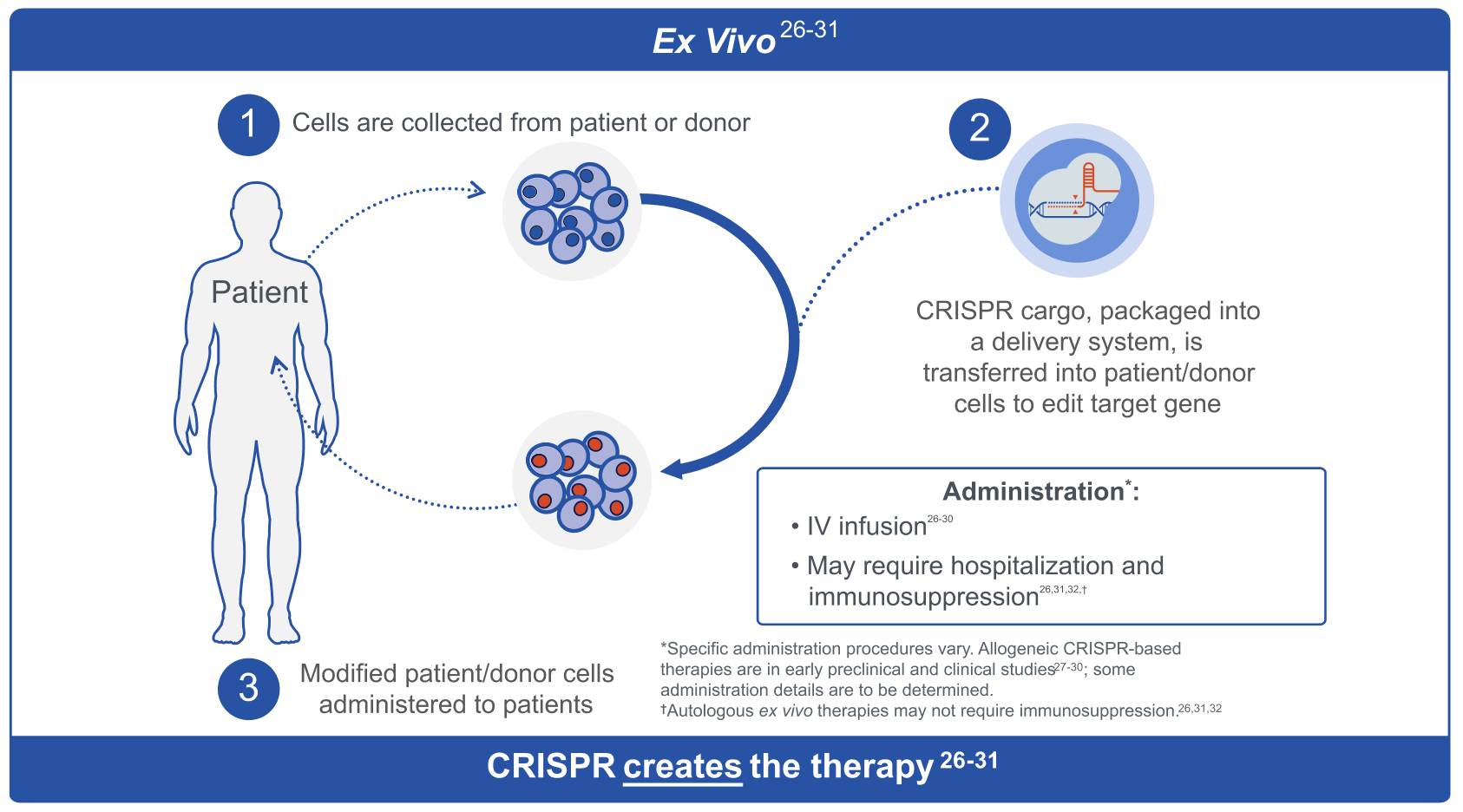
CRISPR, clustered regularly interspaced short palindromic repeats; IV, intravenous.
The History of CRISPR Technology
In 2012, scientists demonstrated CRISPR’s potential as a programmable gene editing tool, successfully editing DNA in human cells.33 Following thorough preclinical and clinical studies, the first ex vivo CRISPR-based therapy received FDA approval in 2023.26 Ongoing Phase 3 clinical trials are investigating the safety and efficacy of in vivo CRISPR therapies.34-36
The Journey of CRISPR: From Discovery to Innovation
Cas9, CRISPR-associated protein 9; CRISPR, clustered regularly interspaced short palindromic repeats; FDA, US Food and Drug Administration; SCD, sickle cell disease; TDT, transfusion-dependent β-thalassemia.
Connect With Intellia
Our goal is to equip healthcare professionals with the essential knowledge to understand the science of CRISPR and its potential as a therapeutic option to support informed decision-making.
Please submit any specific questions here:
References
- Stern LK, Patel J. Methodist Debakey Cardiovasc J. 2022;18(2):59-72.
- Smith TD, Riedl MA. Ann Allergy Asthma Immunol. 2024;133(4):380-390.
- Riedl MA, et al. J Allergy Clin Immunol Pract. 2024;12(4):911-918.
- Fijen LM, et al. N Engl J Med. 2022;386(11):1026-1033.
- Lauffer MC, et al. Commun Med (Lond). 2024;4(1):6.
- Inclisiran. Package insert. Novartis; 2024.
- Givosiran. Package insert. Alnylam Pharmaceuticals; 2024.
- Liu F, et al. MedComm (2020). 2024;5(9):e645.
- Longhurst HJ, et al. N Engl J Med. 2024;390(5):432-441.
- Gillmore JD, et al. N Engl J Med. 2021;385(6):493-502.
- Doudna JA. Nature. 2020;578(7794):229-236.
- Hsu PD, et al. Cell. 2014;157(6):1262-1278.
- Ay C, Reinisch A. Wien Klin Wochenschr. 2024;137(9-10):261-271.
- Bergendahl LT, et al. Protein Sci. 2019;28(8):1400-1411.
- Saito Y, et al. Int J Mol Sci. 2021;23(1):25.
- Bezerra F, et al. Front Mol Neurosci. 2020;13:592644.
- Caccia S, et al. Pediatr Allergy Immunol Pulmonol. 2014;27(4):159-163.
- Hillary VE, Ceasar SA. Mol Biotechnol. 2023;65(3):311-325.
- Press release. The Nobel Prize in Chemistry 2020. October 7, 2020. Accessed March 12, 2025. https://www.nobelprize.org/prizes/chemistry
/2020/press-release/ - Gostimskaya I. Biochemistry (Mosc). 2022;87(8):777-788.
- Ishino Y, et al. J Bacteriol. 1987;169(12):5429-5433.
- Our Science. Intellia Therapeutics. Accessed March 12, 2025. https://www.intelliatx.com/our-science/
- Gillmore JD, et al. Protocol. N Engl J Med. 2021;385(6):493-502.
- Longhurst HJ, et al. Protocol. N Engl J Med. 2024;390(5):432-441.
- Li Y, et al. Biomaterials. 2020;234:119711.
- Exagamglogene autotemcel. Package insert. Vertex Pharmaceuticals Incorporated; 2023.
- ClinicalTrials.gov identifier: NCT05565248. Updated May 23, 2024. Accessed December 14, 2025. https://clinicaltrials.gov/study/NCT05565248
- ClinicalTrials.gov identifier: NCT05885464. Updated January 13, 2025. Accessed December 14, 2025. https://clinicaltrials.gov/study/NCT05885464
- ClinicalTrials.gov identifier: NCT05643742. Updated November 14, 2025. Accessed December 14, 2025. https://clinicaltrials.gov/study/NCT05643742
- ClinicalTrials.gov identifier: NCT05795595. Updated November 26, 2025. Accessed December 14, 2025. https://clinicaltrials.gov/study/NCT05795595
- Martínez Bedoya D, et al. Front Immunol. 2021;12:640082.
- Li H, et al. Signal Transduct Target Ther. 2020;5(1):1-23.
- Jinek M, et al. Science. 2012;337(6096):816-821.
- Jinek M, et al. Elife. 2013;2:e00471.
- Press release. Intellia Therapeutics. October 18, 2023. Accessed March 12, 2025. https://ir.intelliatx.com/news-releases/news-release-details/intellia-therapeutics-announces-fda-clearance-investigational-0
- Press release. Intellia Therapeutics. October 7, 2024. https://ir.intelliatx.com/news-releases/news-release-details/intellia-therapeutics-announces-initiation-haelo-phase-3-study
- Press release. Intellia Therapeutics. November 7, 2024. https://ir.intelliatx.com/news-releases/news-release-details/intellia-therapeutics-announces-third-quarter-2024-financial
- Press Release. CRISPR Therapeutics. February 25, 2019. Accessed September 23, 2025. https://ir.crisprtx.com/news-releases/news-release-details/crispr-therapeutics-and-vertex-announce-progress-clinical
- Jakobsen RS. First Chinese CRISPR gene therapy trial demonstrates safety. CRISPR Medicine News. April 28, 2020. Accessed November 11, 2025. https://crisprmedicinenews.com/news/first-chinese-crispr-gene-therapy-trial-demonstrates-safety/
- Press Release. Intellia Therapeutics. November 9, 2020. Accessed November 11, 2025. https://ir.intelliatx.com/news-releases/news-release-details/intellia-therapeutics-doses-first-patient-landmark-crisprcas9


