To stay up to date on the latest news about Intellia’s technology, pipeline, data releases, and congress presence, sign up to receive email updates from Intellia Medical Affairs.
Want More? Connect With Us!
To stay up to date on the latest news about Intellia’s technology, pipeline, data releases, and congress presence, sign up to receive email updates from Intellia Medical Affairs.
Scientific Exploration in Transthyretin Amyloidosis
About Transthyretin Amyloidosis
Transthyretin amyloidosis (ATTR) is a progressive and often fatal disease,2-4 caused by the misfolding of transthyretin (TTR) protein into amyloid fibrils.5 Aggregation of these amyloid fibrils results in ATTR with cardiomyopathy (CM) and/or polyneuropathy (PN).6,7
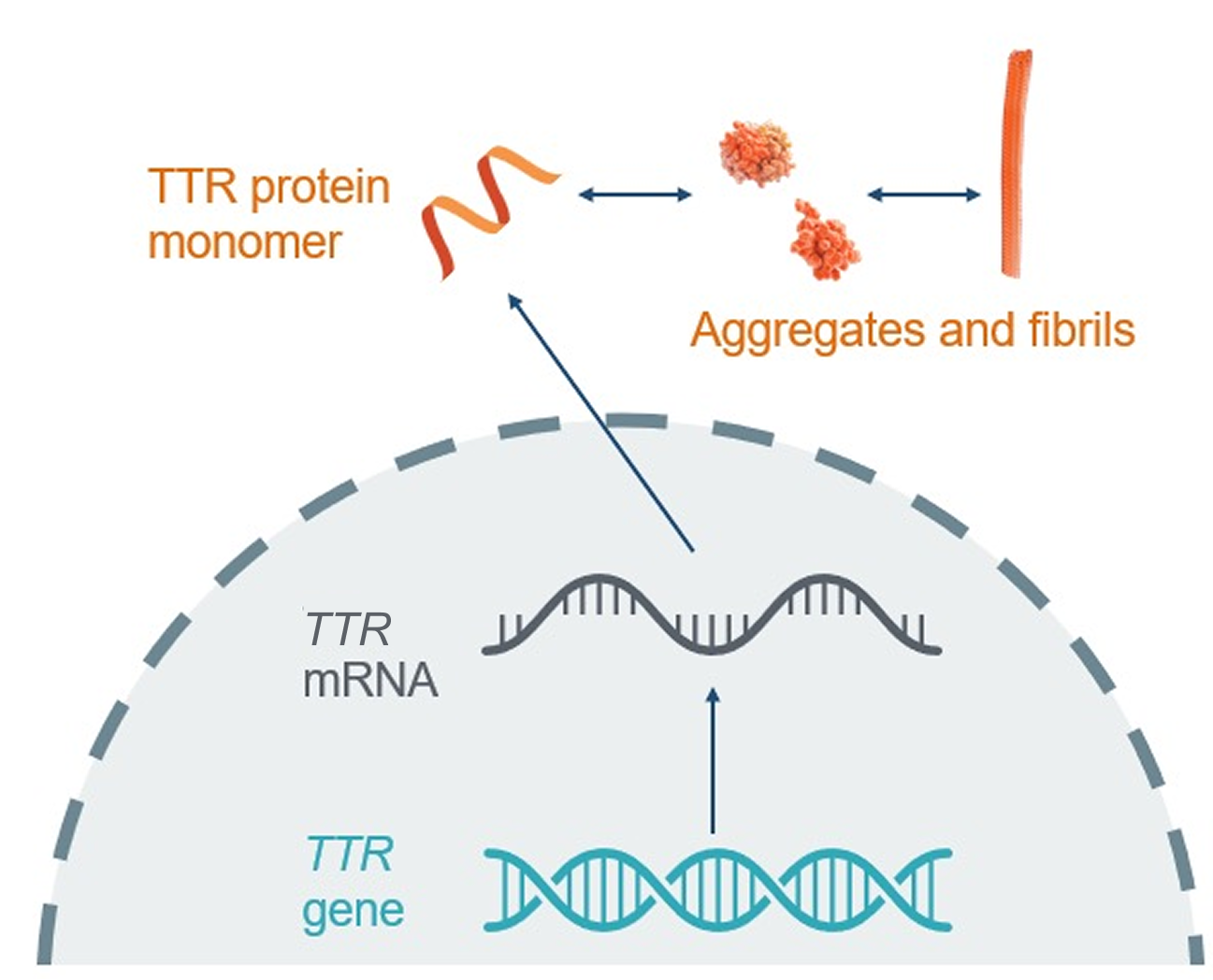
mRNA, messenger RNA; TTR, transthyretin; TTR, transthyretin gene that encodes transthyretin protein.
Therapeutic Landscape
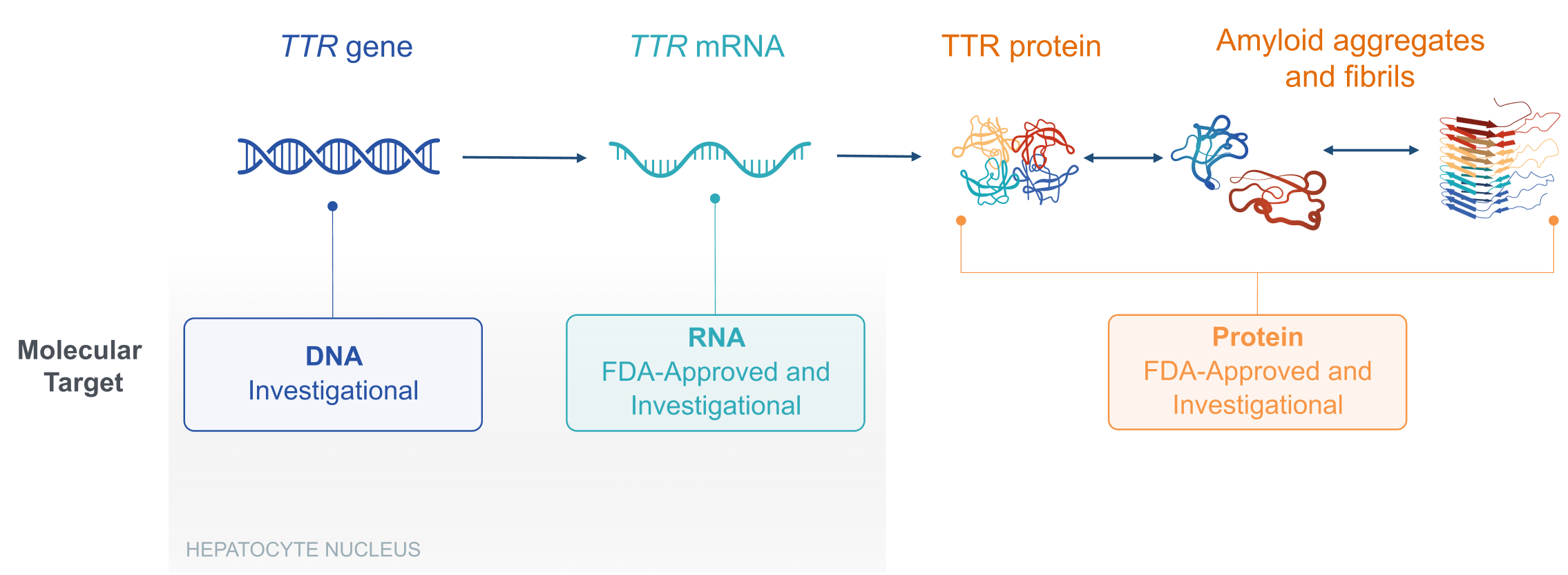
DNA, deoxyribonucleic acid; FDA, US Food and Drug Administration; mRNA, messenger RNA; RNA, ribonucleic acid; TTR, transthyretin; TTR, transthyretin gene that encodes transthyretin protein.
Rationale for CRISPR Research
ATTR is a monogenic disease with both hereditary and acquired forms resulting from TTR protein misfolding1,5:
- Hereditary ATTR: mutation in the gene that codes for TTR protein leads to misaggregation of TTR protein
- Acquired ATTR: spontaneous, nongenetic misaggregation of wild-type TTR protein
Treatment innovation for ATTR continues to progress with multiple investigational therapy studies that target the root cause of disease.9-16 ATTR may be a good therapeutic target for CRISPR-based gene editing because it involves a single gene (TTR) and TTR protein is primarily produced in the liver.1
Connect With Intellia
Our goal is to equip healthcare professionals with the essential knowledge to understand the science of CRISPR and its potential as a therapeutic option to support informed decision-making.
Please submit any specific questions here:
References
- Gillmore JD, et al. N Engl J Med. 2021;385(6):493-502.
- Nativi-Nicolau J, et al. ESC Heart Fail. 2021;8(5):3875-3884.
- Kittleson MM, et al. J Am Coll Cardiol. 2023;81(11):1076-1126.
- Adams D, et al. J Neurol. 2021;268(6):2109-2122.
- Tschöpe C, Elsanhoury A. J Clin Med. 2022;11(8):2148.
- Witteles RM, et al. JACC Heart Fail. 2019;7(8):709-716.
- Shin SC, Robinson-Papp J. Mt Sinai J Med. 2012;79(6):733-748.
- Vutrisiran. Package insert. Alnylam Pharmaceuticals; 2025.
- ClinicalTrials.gov identifier: NCT04601051. Updated May 1, 2025. Accessed December 14, 2025. https://clinicaltrials.gov/study/NCT04601051
- ClinicalTrials.gov identifier: NCT06128629. Updated December 2, 2025. Accessed December 14, 2025. https://clinicaltrials.gov/study/NCT06128629
- ClinicalTrials.gov identifier: NCT06539208. Updated August 6, 2024. Accessed December 14, 2025. https://clinicaltrials.gov/study/NCT06539208
- ClinicalTrials.gov identifier: NCT04153149. Updated November 20, 2025. Accessed December 14, 2025. https://clinicaltrials.gov/study/NCT04153149
- ClinicalTrials.gov identifier: NCT03860935. Updated June 27, 2024. Accessed December 14, 2025. https://clinicaltrials.gov/study/NCT03860935
- ClinicalTrials.gov identifier: NCT06183931. Updated November 17, 2025. Accessed December 14, 2025. https://clinicaltrials.gov/study/NCT06183931
- ClinicalTrials.gov identifier: NCT06260709. Updated November 25, 2025. Accessed December 14, 2025. https://clinicaltrials.gov/study/NCT06260709
- ClinicalTrials.gov identifier: NCT04360434. Updated November 11, 2025. Accessed December 14, 2025. https://clinicaltrials.gov/study/NCT04360434
- Press release. YolTech Therapeutics. July 11, 2024. Accessed March 11, 2025. https://www.yoltx.com/news/press-release/65
- Press release. Alnylam Pharmaceuticals. October 9, 2024. Accessed March 11, 2025. https://investors.alnylam.com/press-release?id=28446
- Press release. BridgeBio Pharma. February 5, 2024. Accessed March 11, 2025. https://investor.bridgebio.com/news-releases/news-release-details/bridgebio-pharma-announces-us-food-and-drug-administration-fda
- Fontana M, et al. Eur Heart J. 2022;43(suppl 2):ehac544.1767.