To stay up to date on the latest news about Intellia’s technology, pipeline, data releases, and congress presence, sign up to receive email updates from Intellia Medical Affairs.
Want More? Connect With Us!
To stay up to date on the latest news about Intellia’s technology, pipeline, data releases, and congress presence, sign up to receive email updates from Intellia Medical Affairs.
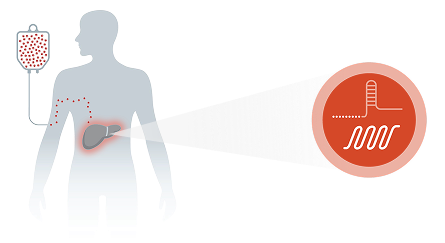
Intellia’s In Vivo Approach
Intellia’s CRISPR-based investigational therapies were designed to precisely edit DNA and halt production of proteins associated with a disease via a one-time infusion.1,2
Intellia’s investigational CRISPR technology is packaged within lipid nanoparticles (LNPs), a nonviral delivery system that transports the CRISPR technology to the desired organ (ie, liver).1,2
How Intellia’s Investigational CRISPR Technology Is Designed to Work1-6

In vivo administration enables a one-time infusion for CRISPR-based gene editing to occur1,2
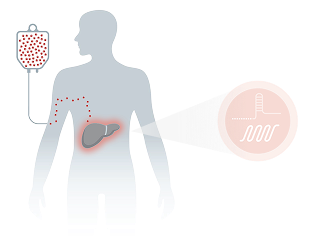

LNP is a nonviral delivery system that distributes preferentially to the liver, where the majority of the target protein is synthesized1-5

A guide RNA directs CRISPR to enable a precise edit in the target gene to potentially inactivate it1,2


The edit is expected to be permanent even after the CRISPR-based therapeutic is cleared from the system1,2,6



Cas9, CRISPR-associated protein 9; CRISPR, clustered regularly interspaced short palindromic repeats; LNP, lipid nanoparticle; RNA, ribonucleic acid.
Two Key CRISPR Components1,7
Intellia’s CRISPR has two components, a guide RNA and Cas9 enzyme, that are designed to target a specific location on the gene containing the disease-associated protein blueprint so that a precise edit can be made.1,7
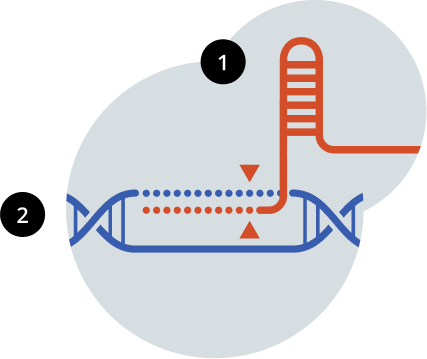

What is guide RNA?
Guide RNA, also called gRNA, acts as a “molecular GPS” for the Cas9 enzyme, helping to locate the precise spot in the DNA that needs editing.1,7

What is Cas9?
Cas9 is the enzyme responsible for editing the gene.1,7
Learn more about the components of CRISPR that aid in its precision by watching the following video:
CRISPR/Cas9 Process
A guide RNA, which acts as a “molecular GPS,” can be designed to target a specific gene of interest. Furthermore, guide RNAs can be carefully engineered to precisely target a very specific location within a gene, with the aim to minimize off-target edits.1,2,7
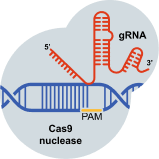


Cas9 first identifies the protospacer adjacent motif (PAM) in the DNA, which allows initial binding.8,9
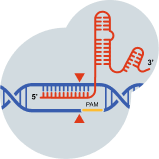


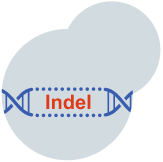


Once activated, the Cas9 nuclease will create a precise double-strand break (DSB) in the target gene, potentially resulting in small insertions and/or deletions (indels) and inactivating the gene.1,8,9



This is intended to stop production of the protein associated with a disease.1
Cas9, CRISPR-associated protein 9; DNA, deoxyribonucleic acid; gRNA, guide RNA; PAM, protospacer adjacent motif.
Natural Repair Process
DNA is naturally prone to spontaneous double-strand breaks (DSB), which can occur randomly in cells 10-50 times per day per cell during standard DNA replication or in response to external stressors such as radiation.10,11 Because these double-strand breaks are so common, the body has natural repair processes to address them.10,11 During this process, small insertions or deletions in DNA may naturally occur, which can disrupt a gene’s instructions for making a particular protein.1,12
CRISPR leverages this process by creating a single precise double-strand break at a specific location in the gene of interest, directed by guide RNA, to activate the body’s natural repair process.1,2,11,12 The use of a guide RNA maximizes precision. As a result, the gene may be permanently inactivated and the disease addressed at the genetic level.1,2
CRISPR, clustered regularly interspaced short palindromic repeats; DNA, deoxyribonucleic acid.
CRISPR technology is being investigated to treat both genetically inherited diseases and acquired diseases that arise from an aberrantly behaving wild-type protein.1,2
Intellia’s Proprietary Lipid Nanoparticle Delivery System
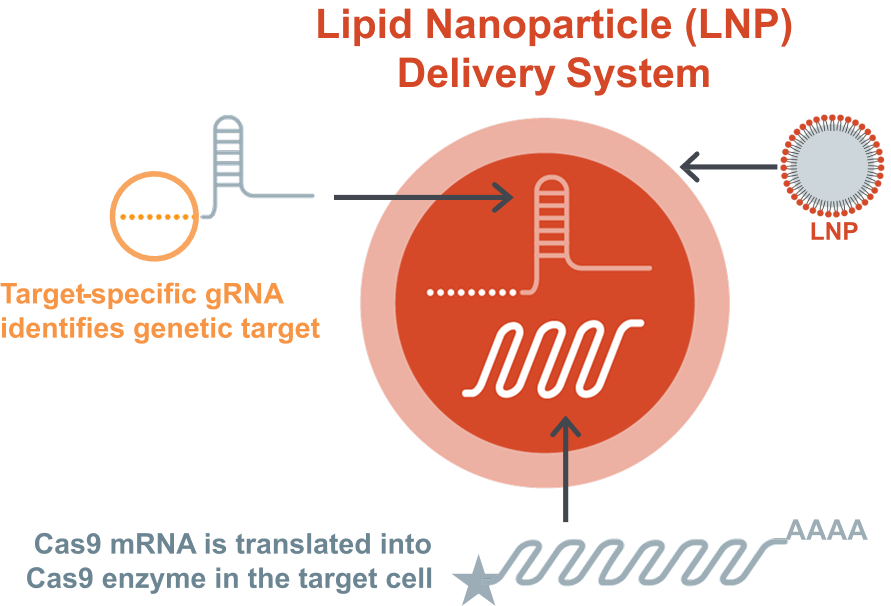
Intellia has a proprietary delivery method designed to enable direct delivery of the LNP and its components to the liver.1 The liver is a clinically relevant target for a range of diseases because it produces many proteins of interest.1
Intellia is studying the use of CRISPR to treat diseases where the liver is the primary production site of the protein of interest, and has developed an investigational approach designed to be a one-time treatment.1,2,14
The precision editing components (ie, guide RNA and Cas9 messenger RNA) are packaged into a proprietary lipid nanoparticle (LNP) delivery system.1,2 Based on a preclinical study, lower immunogenicity has been observed with the nonviral LNP delivery system compared with a viral vector.4
The proprietary lipid nanoparticle delivery system was designed to help maximize CRISPR gene editing efficacy and minimize systemic toxicity through high liver affinity and reduced risk of uptake by other organs.1 Furthermore, the LNP has been shown in preclinical studies to be cleared from circulation and the liver within 5 days, with a goal to reduce the risk of systemic toxicity or immunogenicity.6,*
*These data are from an animal model; the relevance to humans has not been established.
Cas9, CRISPR-associated protein 9; gRNA, guide RNA; mRNA, messenger RNA.
Proposed Mechanism of Action of In Vivo CRISPR-Based Therapies1-3
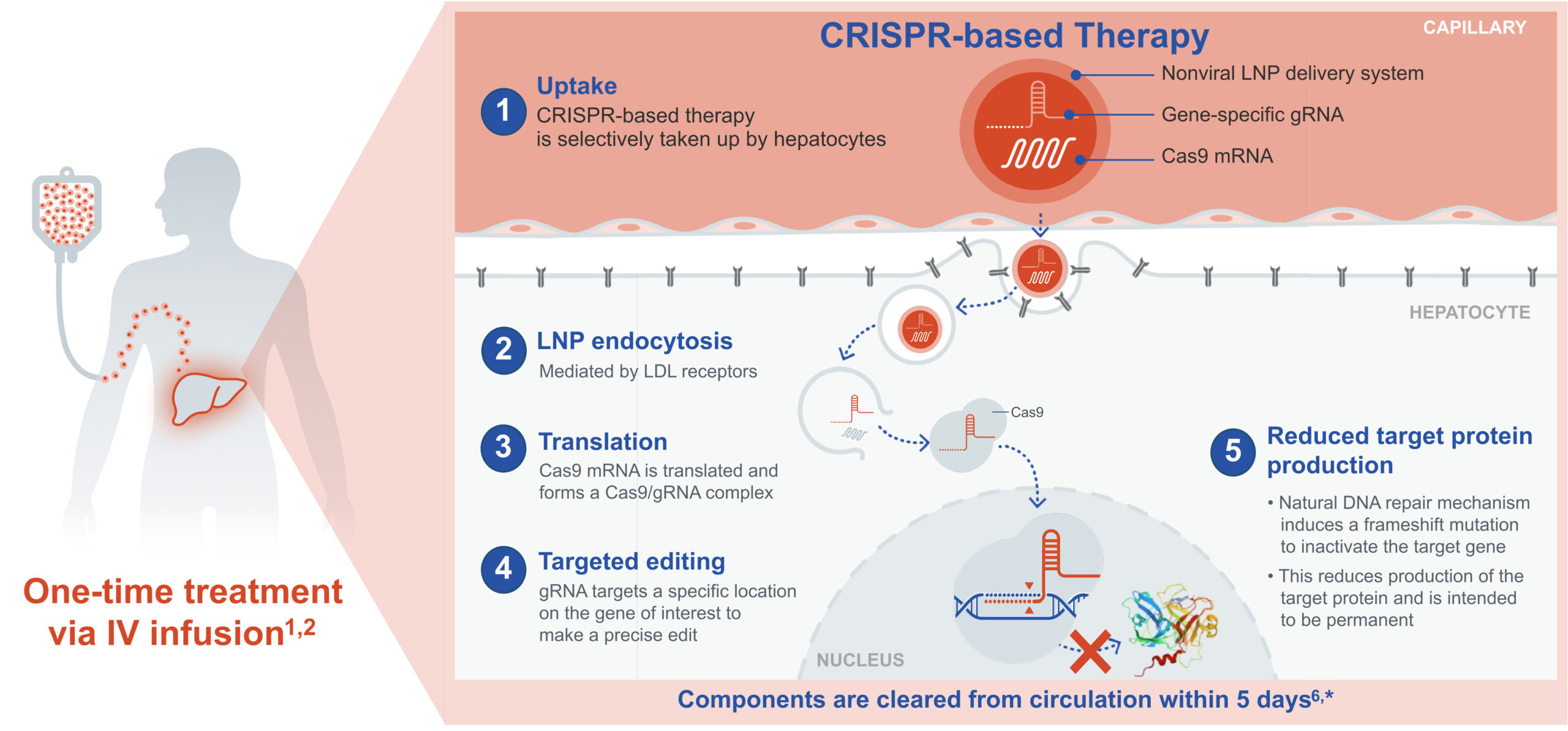
*These preclinical studies were conducted in nonhuman primates and supported the initiation of Intellia’s clinical trials to evaluate the safety and efficacy of its investigational products. The relevance to humans has not been established.
Cas9, CRISPR-associated protein 9; CRISPR, clustered regularly interspaced short palindromic repeats; DNA, deoxyribonucleic acid; gRNA, guide RNA; IV, intravenous; LDL, low-density lipoprotein; LNP, lipid nanoparticle; mRNA, messenger RNA.
In this illustration of the proposed mechanism of action, the LNP is taken up by hepatocytes after a one-time infusion. The CRISPR complex forms in the nucleus of hepatocytes, where it makes a precise edit in the gene of interest.1,2,7 If successful, this edit halts production of the protein linked to the disease.1,2
The edit is expected to be permanent after Intellia's investigational CRISPR-based therapy is cleared from the system.2,6 The guide RNA, which is required for the CRISPR complex to make an edit, shows decreasing plasma levels over time, suggesting that the guide RNA does not persist long term.7,15
Intellia’s In Vivo Approach to Gene Editing
Responsible Innovation: High Precision, High Accuracy
Intellia is committed to improving patient outcomes by developing the most precise CRISPR-based therapies possible through responsible innovation. One way we do this is by thoroughly testing our guide RNAs.16-18 Guide RNAs are the “molecular GPS” that helps the CRISPR complex target the intended location in a gene.7




Learn more about our approach to responsible innovation in CRISPR/Cas9 by watching the following video:
Connect With Intellia
References
- Gillmore JD, et al. N Engl J Med. 2021;385(6):493-502.
- Longhurst HJ, et al. N Engl J Med. 2024;390(5):432-441.
- Longhurst HJ, et al. Supplementary appendix. N Engl J Med. 2024;390(5):432-441.
- Kenjo E, et al. Nat Commun. 2021;12(1):7101.
- Gertz MA, et al. J Am Coll Cardiol. 2015;66:2451-2466.
- Wood K, et al. Presented at: 2nd European Congress for ATTR Amyloidosis; September 1-3, 2019; Berlin, Germany.
- Hillary VE, Ceasar SA. Mol Biotechnol. 2023;65(3):311-325.
- Sternberg SH, et al. Nature. 2015;527(7576):110-113.
- Jiang F, Doudna JA. Annu Rev Biophys. 2017;46:505-529.
- Lieber MR, et al. Subcell Biochem. 2010;50:279-296.
- Cannan WJ, Pederson DS. J Cell Physiol. 2016;231(1):3-14.
- Stinson BM, Loparo JJ. Annu Rev Biochem. 2021;90:137-164.
- Li T, et al. Signal Transduct Target Ther. 2023;8(1):36.
- Intellia Company Overview. Intellia Therapeutics. August 2024.
- Intellia Therapeutics. Data on file.
- O’Connell DJ, et al. Presented at: European Society of Gene and Cell Therapy 27th Annual Meeting; October 23, 2019; Barcelona, Spain.
- Patel N, et al. Presented at: 23rd Annual Meeting of the American Society of Gene and Cell Therapy; May 12, 2020; Los Angeles, CA, and virtual.
- O’Connell DJ. Presented at: 24th Annual Meeting of the American Society of Gene and Cell Therapy; May 10, 2021; virtual congress.
- Sepp-Lorenzino L. Presented at: US Hereditary Angioedema Association; June 11, 2024; Fairfax City, VA.
- Gillmore JD, et al. Supplementary appendix. N Engl J Med. 2021;385(6):493-502.



